At Compound, we’re obsessed with faster iterations for drug discovery. We’ve written about this and our frustration with feedback loops led us to publish a Playbook for Human Evidence last year. This playbook highlights eight ways that drug hunters can try their drugs as quickly in humans as possible. This helped more founders and operators than we could have imagined so wanted to expand on each of the eight pathways.
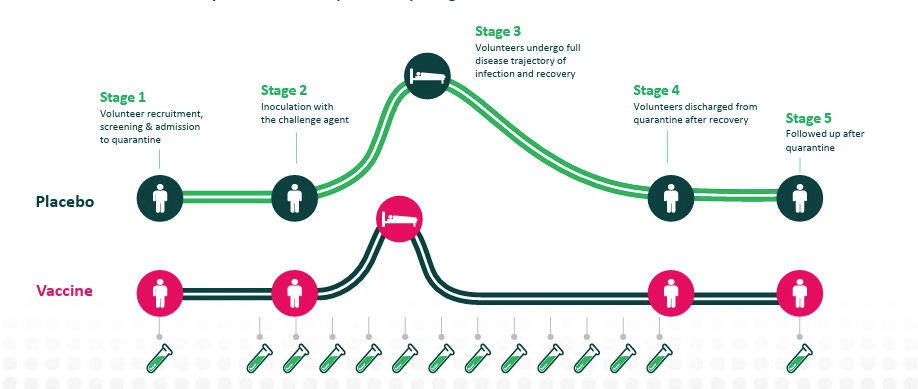
One pathway of fast human efficacy data is human challenge trials (HCTs). HCTs test experimental therapeutics and vaccines by intentionally infecting healthy volunteers. These radical trials are regularly conducted in the infectious disease vertical to test drug efficacy before underwriting large Phase III trials. In some cases, like Cholera, HCTs are enough in themselves for FDA approval of new drugs. Despite the frequent use of HCTs, we find many early-stage founders are unaware of this route for human efficacy evidence. With this piece we go through several case studies of HCTs and how they’ve sped up the drug discovery process and a little-known public company, hVIVO, the main engine of HCTs.
HCTs aren’t new and for centuries the west has been toying with challenge studies as they tried to understand how to protect people from smallpox, malaria, syphilis, typhoid, gonorrhea and other diseases. Challenge studies also have an ethically fraught past where children, those economically disadvantaged, or imprisoned were initial subjects of HCTs. In fact, a smallpox HCT in 1721 offered its six prisoners pardons after taking part in the trial.
The first accredited HCT was in 1796 where scientist Edward Jenner transferred cowpox pus from a milkmaid’s hand and scratched it into the arm of an 8-year-old boy named James Phipps. Weeks later Jenner intentionally exposed the boy, James Phipps, to smallpox. Astoundingly, James did not get sick. This caused a surge in cowpox vaccinations all over the UK, US, South America India, saving millions of people. In fact Jenner “shipped [the cowpox] vaccine to President Thomas Jefferson, who had eighteen of his relatives vaccinated and established the National Vaccine Institute…”. James Phipps was exposed over his lifetime to smallpox (which he never caught) by Jenner and was eventually given a cottage by Jenner as a small thanks to his contribution.
Since then, over 280 HCTs have been conducted on non-lethal, infectious diseases. HCTs are frequently used by infectious disease programs because the controlled trial environment allows for much fewer trial participants (50 or less), constricted timelines (because infections are controlled vs. Phase IIIs where participants wait to get ill), all resulting in materially less cost. Where Phase IIIs for common inflections such as influenza and COVID-19 cost $100M+, the reported rate for efficacy readouts for HCTs is on the order of mid-seven figures to low-eight figures.
For startups this price and time differential matters tremendously. The most valuable aspects for founders include:
It’s materially easier to raise $5M vs. $100M. This especially is valuable if you’re a first time founder and have never developed a drug previously. There is a higher burden of proof and being able to prove efficacy with $5M can help in downstream fundraising and reputation building.
If your startup is building a new therapeutic platform for new target discovery or modality, there is a chance your first drug to the clinic doesn’t work. The relative cost of HCTs vs. Phase III’s mean that your company could further other therapeutics and generally have more shots on goal since $100M wasn’t yet spent. This is getting closer to what’s being proposed at “Clinic in the Loop”.
Positive efficacy with HCTs will make raising $100M for a Phase III materially more feasible since the drug/vaccine is already derisked.
In some cases, HCTs alone are enough for FDA approval, drastically improving the ROI for drug development
Some recent examples of drugs that have gone through HCTs include a Cholera vaccine (VaxChora) from Emergent Biosolutions ($EBS). Cholera is especially dangerous because it causes profuse vomiting and diarrhea causing rapid dehydration. This is spread through infected fecal matter in water sources. To combat this, VaxChora was approved with only a human challenge trial. The HCT alone was used as the final endpoint because most Cholera outbreaks are unpredictable making prospective recruiting hard. On the flipside, in places where Cholera is endemic there’s already baseline immunity which would skew the results of the study.
While Cholera is a rare example of approval based on HCTs alone, there’s large scope in the modalities and treatments that have been tested with HCTs.
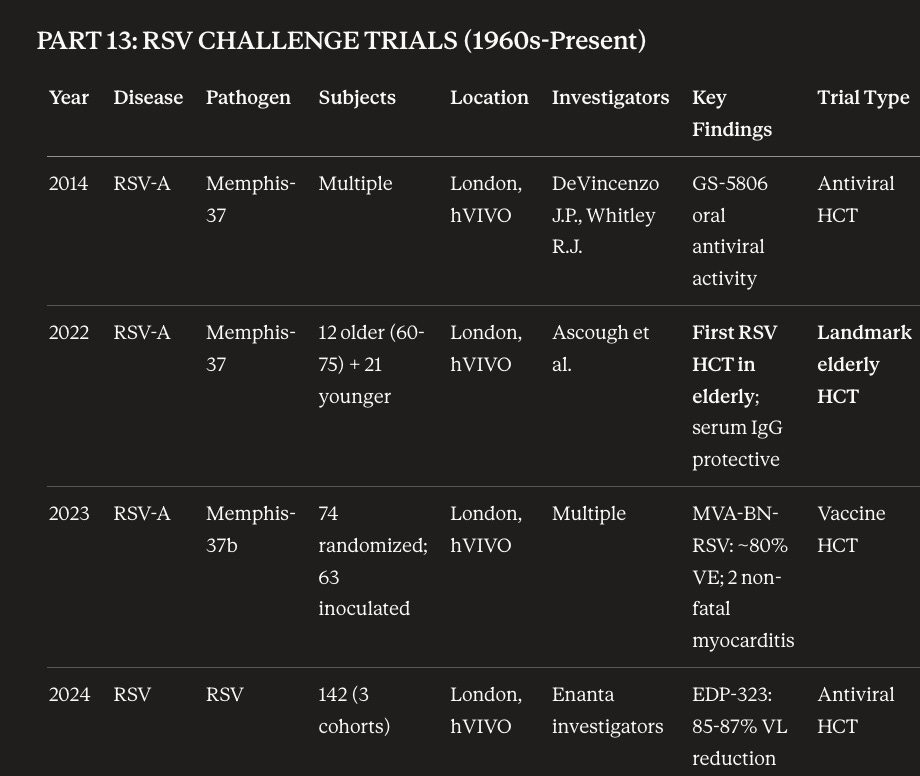
When studying all HCT trials, we see that clinical-stage private companies, big pharma, and everyone in between have conducted HCTs. T-cell priming vaccines for Dengue and COVID-19 by Emergex Vaccines, RSV antivirals by Merck and Enanta, intranasal COVID vaccines by CodaGenix, Malaria vaccines by GSK, oral influenza vaccines by Vaxart are some recently published HCTs. Pfizer also took a new mRNA vaccine for influenza through HCTs. We scraped all historic HCT information together to prompt a fairly comprehensive database of HCTs.
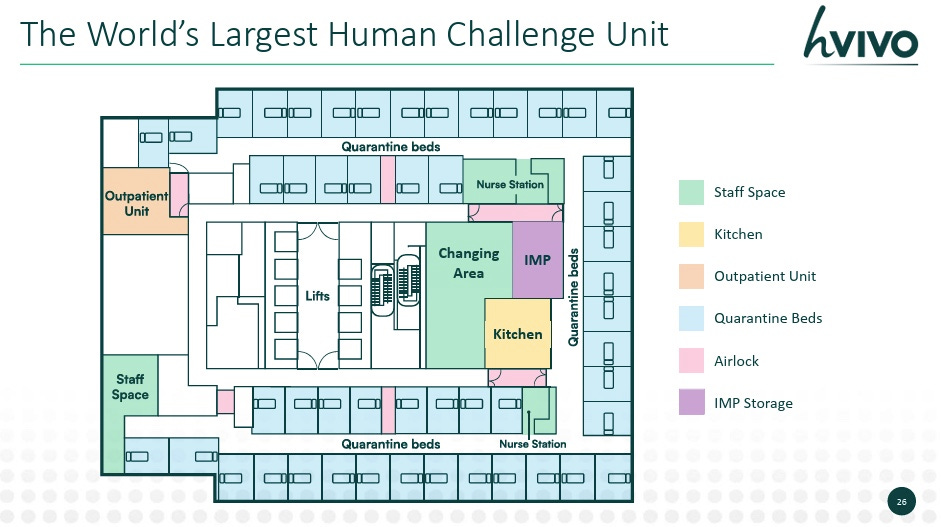
As we’ve catalogued these trials, we’ve seen that most HCTs occur at a few centers including hVIVO, University of Maryland, Radbound University, Oxford Vaccine Group and Johns Hopkins. hVIVO is the only public company that primarily focuses on HCTs and through looking at their filings, we can see some pretty interesting details including $63M projected income for 2025. Additionally, hVIVOs reports highlight little discussed/niche complexities such as viral/bacterial manufacturing, containment, and HCT setup. To understand what the relatively small state of human challenge trials is, we need only look at hVIVO’s largest HCT facility below (which isn’t that big!). HCTs are in this liminal space where they’re industry validated, used extensively in infectious disease but not widely enough to merit more than one public market contract research organization (CRO).
What we’re excited about is combining all the work on viral and bacterial evolution prediction with predictive HCTs. Put another way, doing HCTs on evolved bacteria and viruses to make preventative therapies and vaccines for next evolutionary steps. In a world with increasing antimicrobial resistance, viral escapes, and potential for prompt engineered viruses, we need to enhance protective methods to lessen global impacts of known near-step mutations.
A broader point is that the concept of HCTs is moving outside of infectious disease. We’ve seen a few examples of companies that want to use the premise of accelerated disease to study interventions in a fast and (somewhat) capital efficient way. In space, astronauts experience muscle and bone wasting much more quickly than on Earth, creating a perfect environment for companies like Ursa to conduct HCTs for a new suites of diseases. Other ideas we’ve discussed are around allergy HCT where people with mild allergies are exposed to allergens under medical supervision and then given a drug. Hypoxia chambers which reduce atmospheric oxygen could allow for the study of COPD, pulmonary hypertension, or angina in healthy subjects to track improvement with a therapeutic intervention vs. not. Peptide CCK-4 is anxiety inducing and has been proposed for decades as an anxiety challenge trial molecule.
In the end, treating illness is a human challenge trial but as humanity pushes towards enhancement, likely the future will be full of challenging our current mental, physical, metabolic states for their optimization.